Energy Conversion
Energy conversion, also known as energy transformation, refers to the process of changing one form of energy into another. This process occurs constantly and in various ways throughout the day. There are numerous forms of energy, including thermal energy, electrical energy, nuclear energy, electromagnetic energy, mechanical energy, chemical energy, and sound energy, among others. The term “energy transformation” is specifically used when energy changes from one form to another. Whether energy is transferred or transformed, the total amount of energy remains constant, in accordance with the law of conservation of energy.
Law of Energy Conversion
The law of energy conversion, also known as the law of conservation of energy, states that energy cannot be created or destroyed; it can only be transformed from one form to another. This fundamental principle applies to all energy conversions. The field of thermodynamics explores how energy is converted from one form to another. The following section provides details on how the laws of thermodynamics apply to energy conversion.
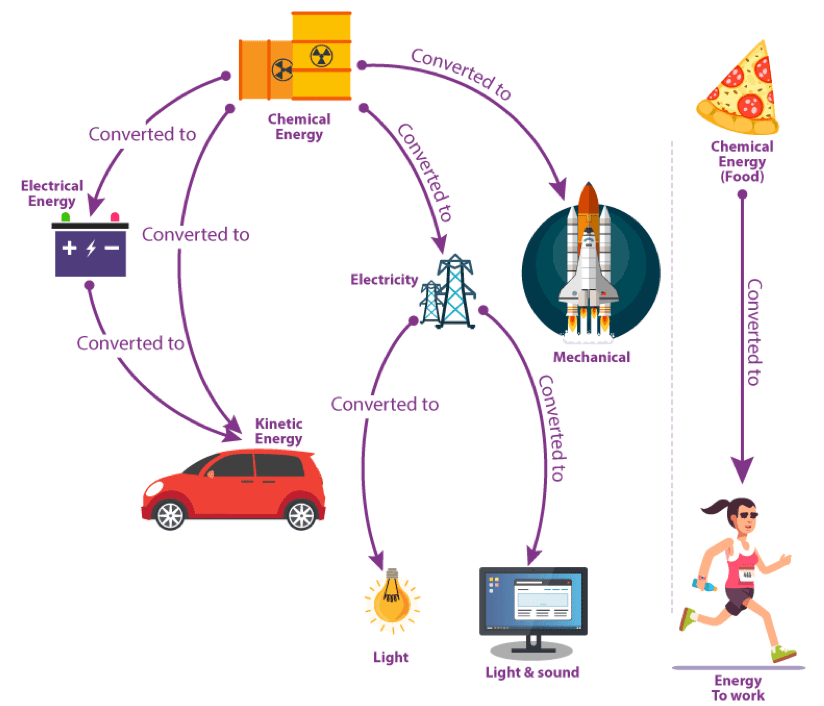
Examples of Energy Conversion Various everyday examples illustrate energy conversion processes. Here are some examples:
- Rubbing both hands together for warmth Energy conversions involved: Kinetic Energy to Thermal Energy
- A falling object speeding up Energy conversions involved: Gravitational Potential Energy to Kinetic Energy
- Using a battery-powered torchlight Energy conversions involved:
- In the battery: Chemical Energy to Electrical Energy
- In the bulb: Electrical Energy to Radiant Energy
- In a Geothermal Power Plant Energy conversions involved: Heat Energy to Electrical Energy
- In a Thermocouple Energy conversions involved: Heat Energy to Electrical Energy
- In Hydroelectric Dams Energy conversions involved: Gravitational Potential Energy to Electric Energy
- In an Electric Generator Energy conversions involved: Kinetic Energy/Mechanical Energy to Electric Energy
- In Windmills Energy conversions involved: Wind Energy to Mechanical Energy or Electric Energy
- In OTEC (Ocean Thermal Energy Conversion) Energy conversions involved: Heat Energy to Electric Energy or Mechanical Energy
- Using a Microphone Energy conversions involved: Sound Energy to Electric Energy
- Photosynthesis in Plants Energy conversions involved: Solar Energy to Chemical Energy
- In Piezoelectrics Energy conversions involved: Strain Energy to Electric Energy
- In an Electric Lamp Energy conversions involved: Electric Energy to Heat Energy and Light Energy
- Burning of Wood Energy conversions involved: Chemical Energy to Heat and Light Energy
- In Fuel Cells Energy conversions involved: Chemical Energy to Electric Energy
- In a Steam Engine Energy conversions involved: Heat Energy to Mechanical Energy
- In an Electric Heater Energy conversions involved: Electric Energy to Heat Energy
Energy Conversion: Transfer and Transformation
Energy Transfer vs Energy Transformation vs Energy Conservation It is essential to understand the distinctions between energy transfer, energy transformation, and energy conservation.
- Energy Transfer: Energy transfer refers to the movement of energy from one location to another. It involves the transfer of energy without changing its form. A common example is the flow of electricity from a wall plug, through a charger, and into a battery.
- Energy Transformation: Energy transformation is the process of changing energy from one type to another. It involves the conversion of energy from one form to a different form. An illustration of this is seen in a hydroelectric dam, where the kinetic energy of water is converted into electrical energy.
- Energy Conservation: Energy conservation refers to the principle that the total amount of energy in a system remains constant. Energy may transfer or transform within the system, but the total energy remains unchanged.
Energy is neither created nor destroyed, it remains constant in the universe. However, energy can change its form and transfer between objects.
A familiar example of energy transfer is the transfer of kinetic energy, associated with motion, from a moving object to a stationary one through work. Work measures energy transfer and refers to the force exerted by an object over a distance. For instance, when a golf club strikes a stationary ball, some of the club’s kinetic energy is transferred to the ball as work is done on it. In such transfers, energy moves between objects but remains in the same form. While kinetic energy transfers are observable, other essential transfers are less apparent.
Thermal energy pertains to the internal energy of a system resulting from its temperature. When a substance is heated, its temperature rises because its molecules move faster and gain thermal energy through heat transfer. Temperature measures the level of hotness or coldness of an object, while heat refers to the transfer of thermal energy from a hotter system to a cooler one. Thermal energy transfers occur through conduction, convection, and radiation.
Conduction occurs when thermal energy is transferred between neighboring molecules in contact with each other. For example, when a metal spoon is placed in boiling water, even the portion not touching the water becomes hot. This is because metal is an efficient conductor, allowing heat to easily travel through the material. The molecular vibrations at the end of the spoon in contact with the water spread throughout the entire spoon, raising the temperature. Materials like wood and plastic, known as insulators, are poor conductors, as heat does not readily travel through them.
Convection only happens in fluids such as liquids and gases. When water is boiled on a stove, the molecules at the bottom of the pot, closest to the heat source, gain thermal energy first. They become faster and spread out, resulting in a lower density at the bottom. These molecules rise to the top, and cooler, denser water takes their place. This cycle creates a current of sinking, heating, rising, cooling, and sinking again.
Radiation, the third type of heat transfer, is vital for life on Earth and plays a role in heating bodies of water. Unlike conduction and convection, radiation does not require direct contact between the heat source and the object being heated. It can transfer heat even through the vacuum of space. The sun is the primary source of thermal energy on Earth, radiating electromagnetic waves, including visible light. Materials on Earth absorb these waves, utilizing them as energy or reflecting them back into space.
During an energy transformation, energy changes its form. For instance, a ball at the top of a hill possesses gravitational potential energy, which arises from its position in a gravitational field and its potential to do work. As the ball is pushed downhill, its potential energy is converted into kinetic energy. The ball continues to lose potential energy and gain kinetic energy until it reaches the bottom of the hill.
In an ideal frictionless scenario, the ball would continue rolling indefinitely, maintaining only kinetic energy. However, on Earth, the opposing force of friction transforms the ball’s kinetic energy into heat, causing it to stop at the bottom of the hill. Just like energy transfers, energy is conserved in transformations.
In nature, energy transfers and transformations occur constantly, as seen in coastal dune environments. When thermal energy from the sun reaches the land and ocean, water, having a high heat capacity, heats up more slowly than land. This temperature difference generates convection currents that manifest as wind.
How do energy transfer and energy transformation differ?
These two concepts can be distinguished quite easily.
Energy transfer refers to the process by which energy, such as heat energy, is transferred from one body to another through a medium. For example, when we transfer kinetic energy from our hand to a ball, it allows the ball to travel a certain distance.
On the other hand, energy transformation refers to the conversion of one form of energy into another. Energy continuously undergoes transformations in a cycle. For instance, when an electrical speaker converts electrical energy into sound energy, it exemplifies energy transformation. Similarly, when we throw a ball upwards, the initial kinetic energy transforms into potential energy as the ball reaches its maximum height. As the ball comes down, the potential energy then converts back into kinetic energy.